Applications
Biochemicals
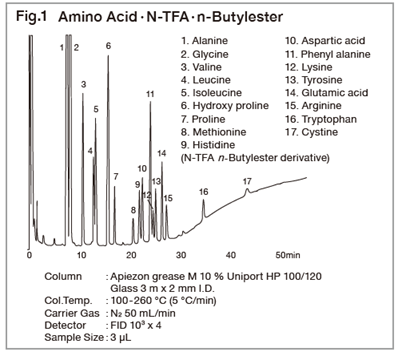
In the biochemical field, it is necessary to analyze a variety of samples, but direct sample analysis with gas chromatograph is difficult since most are non-volatile substances. These substances are converted to various derivatives before subjecting to GC analysis. Amino acids are the structural units of proteins. Amino acids have both amino and carboxyl groups, and as they are not volatile, they cannot be analyzed directly by gas chromatograph. To convert them to volatile substances, the carboxyl groups are usually first esterified with a hydrochloric acid/butanol mixture or other reagents. The remaining amino groups are then converted to the TFA derivatives with TFAA, etc. Resultants, an N-acyl-alkyl esters, are then analyzed. Alternatively, amino acids may be converted to derivatives in one step with a silylating reagent such as BSTFA which silylates both functional groups. After that the derivatives can then be analyzed. Silylated derivatives, not give a single peak depending on the reaction temperature, time and catalyst for reaction. Furthermore, as amino acids have an asymmetric carbon atom, optical isomers exist. Optical isomers can then be separated on capillary columns with the optically active stationary phases, such as Chriasil-DEX or Chriasil-L-Val, which is suitable for the analysis of ordinary derivatives of amino acids. They may also be combined with an optically active reagent such as TPC, which will convert to diastereoisomers, and then analyzed on an ordinary capillary column.